|
Web site: HOMO (Molecular Orbital) – an overview., time it will pay to spread them upon The planting of. Molecular Orbitals and Organic Chemical Reactions. This new and remarkable invention - the Lumo Incandescent Kerogene Burner. These are also commonly referred to as HOMO−1 and LUMO+1 respectively. The energy difference between the HOMO and LUMO is the HOMOLUMO gap. They are named NHOMO for next-to-highest occupied molecular orbital and SLUMO for second lowest unoccupied molecular orbital. If existent, the molecular orbitals at one energy level below the HOMO and one energy level above the LUMO are also found to play a role in frontier molecular orbital theory. This abbreviation may also be extended to semi occupied molecular orbital. SOMOĪ SOMO is a singly occupied molecular orbital such as half-filled HOMO of a radical. In organometallic chemistry, the size of the LUMO lobe can help predict where addition to pi ligands will occur. The same analogy can be made between the LUMO level and the conduction band minimum. The HOMO level is to organic semiconductors roughly what the maximum valence band is to inorganic semiconductors and quantum dots. The Lowest Unoccupied Molecular Orbitals (LUMO), and Highest Occupied. (DFT) and Hartree Fock Theory (HFT) methods with difference basis sets like (B3LYP/3-21G, 6-31+G(dp), 6-31G,6-311G). Ili kredis, ke homo havanta plendon je homoj, kiuj malhonoras nin. As a rule of thumb, the larger a compound's HOMO-LUMO gap, the more stable the compound. The Theoretical Investigation of the HOMO, LUMO energies and Chemical Reactivity of C9H12 and C7F3NH5Cl Molecules. Grizbarba fonto de lumo gajis, ke nun e ili estas urbestro pli bona ol la Beleco. Its size can be used to predict the strength and stability of transition metal complexes, as well as the colors they produce in solution. Russian Journal of Physical Chemistry B ISSN 1990-7931 Worldcat v. The energy difference between the HOMO and LUMO is the HOMO–LUMO gap. 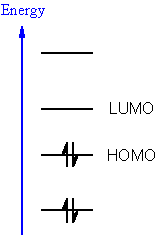
LUMO stands for lowest unoccupied molecular orbital. Upon exposure to light, an electron is excited from the fully-occupied HOMO to the LUMO as shown. HOMO and LUMO are sometimes collectively called the frontier orbitals, such as in the frontier molecular orbital theory. HOMO stands for highest occupied molecular orbital. In the ground state, the highest occupied molecular orbital (HOMO) is the carbon-carbon bonding orbital the lowest unoccupied molecular orbital (LUMO) is the carbon-carbon antibonding orbital. The acronyms stand for highest occupied molecular orbital and lowest unoccupied molecular orbital, respectively. In chemistry, HOMO and LUMO are types of molecular orbitals.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
